|
“Multiple sclerosis is arguably the most complex disease ever described,” says Sergio Baranzini, a geneticist at the University of California, San Francisco. The drugs only reduce the number and severity of flare-ups and, in some cases, slow the visible marks of brain damage. In fact, none of the 15 FDA-approved drugs for MS, which all modify or suppress the immune system, actually stop the disease. Ocrevus isn’t a cure, and it offers no relief for 30 to 40 percent of patients with primary progressive MS. 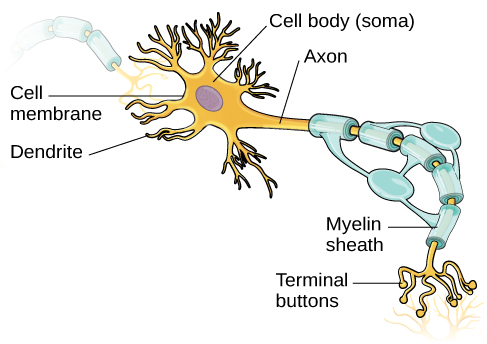
With that hopeful note, though, comes frustration. 
Now, nearly a quarter century later, there’s something that helps some people with the most aggressive form of the disease. The first drug to treat relapsing-remitting MS came on the market in 1993, Lublin notes. “We finally have an approved therapy for primary progressive MS,” says Fred Lublin, a neurologist at the Icahn School of Medicine at Mount Sinai in New York City. Food and Drug Administration approved it as a treatment for primary progressive and relapsing-remitting MS. Ocrelizumab was heralded as a breakthrough, and in March, the U.S. Over a year’s time, these patients experienced about half as many flare-ups as those taking another commonly prescribed drug, a different research group reported in the same issue of the journal. The drug also helped people with relapsing-remitting MS, which, as the name implies, includes shifts between disability and wellness. Brain scans of patients with primary progressive MS showed fewer signs of damage and the patients’ ability to walk deteriorated more slowly than in individuals who received a placebo, researchers reported in January in the New England Journal of Medicine. The drug also held promise for patients with the other major form of the disease, relapsing-remitting MS, which afflicts about 340,000 people nationwide.įor some people, ocrelizumab seemed to work. Early research suggested the drug could help some of the roughly 60,000 people in the United States, like Davis, suffering from primary progressive MS. The drug offered a first sliver of hope for patients waiting for a cure, or at least something to slow down the disease’s staggering march. He can no longer get up to go to the bathroom or grab a snack from the fridge.ĭavis hoped life might improve when he was chosen in 2012 to participate in a clinical trial of a drug called ocrelizumab. Diagnosed in May 2011, Davis relied on a wheelchair within six months. He has the most severe form of multiple sclerosis, known as primary progressive MS, a worsening disease that destroys the central nervous system. Today, the 48-year-old from Albuquerque barely gets out of bed. :max_bytes(150000):strip_icc()/neuron-anatomy-58530ffe3df78ce2c34a7350.jpg)
He surfed, hiked, skateboarded and rock climbed. James Davis used to be an avid outdoorsman.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
